Andhra Pradesh BIEAP AP Inter 2nd Year Chemistry Study Material Lesson 6(c) Group-17 Elements Textbook Questions and Answers.
AP Inter 2nd Year Chemistry Study Material Lesson 6(c) Group-17 Elements
Very Short Answer Questions
Question 1.
Which halogen produces O2 and O3 on passing through water?
Answer:
Fluorine produces O2 and O3 on passing through water.
3F2 + 3H2O → 6HF + O3
2F2 + 2H2O → 4HF + O2
Question 2.
Interhalogen compounds are more reactive than the constituent halogens except fluorine – explain.
Answer:
Inter halogen compounds are more reactive than halogens. This is because X – X’ bond in interhalogens is weaker than X – X bond in halogens except F – F bond.
![]()
Question 3.
What is the use of ClF3?
Answer:
ClF3 is very useful fluorinating agent and it is used for the production of VF6 in the enrichment
of U235.
U(s) + 3ClF3(l) → UF6(g) + 3ClF(g)
Question 4.
Write two uses of ClO2.
Answer:
Uses of ClO2:
- ClO2 is highly reactive oxidising agent.
- It is used as bleaching agent for paper pulp and textiles.
- It is used in water treatment.
Question 5.
Why are halogens coloured?
Answer:
Halogens are coloured due to the absorption of radiations in visible region which results in the excitation of outer electrons to higher energy level. Halogens absorb different quanta of radiation and display different colours.
Question 6.
Write the reactions of F2 and Cl2 with water. [Mar. 14]
Answer:
Fluorine produces O2 and O3 on passing through water.
3F2 + 3 H2O → 6HF + O3
2F2 +2 H2O → 4HF + O2
Chlorine dissolves in water giving a solution of chlorine water. A freshly prepared solution of chlorine water contains HCl and HOCl .
Cl2 + H2O → HCl + HOCl
HOCl is unstable and dissociates to give nascent oxygen
HOCl → HCl + (O)
![]()
Question 7.
With which neutral molecule, ClO– is isoelectronic ? Is that molecule a Lewis base ? (Hint: ClF; Yes)
Answer:
- ClO– is isoelectronic with the neutral molecule ClF.
- Yes. ClF is a Lewis base (Electron pair donar).
Question 8.
Arrange the following in the order of the property indicated for each set.
a) F2, Cl2, Br2, I2 – increasing bond dissociation enthalpy.
b) HF, HCl, HBr, HI – increasing acidic strength
c) HF, HCl, HBr, HI – increasing boiling points.
Answer:
a) Increasing bond’ dissociation enthalpy order
I2 < F2 < Br2 < Cl2
b) Increasing acidic strength order
HF < HCl < HBr < HI
c) Increasing boiling points order
HCl < HBr < HI < HF
Question 9.
Electron gain enthalpy of fluorine is less than that of chlorine – explain.
Answer:
The negative electron gain enthalpy of the fluorine is less them that of chlorine. This is due to small size of fluorine atom which results in strong inter electronic repulsions in the relatively small 2p orbitals of fluorine and thus the incoming electron does not experience much attraction.
Question 10.
HF is a liquid while HCl is a gas – explain.
Answer:
HF is a liquid due to the presence of inter molecular hydrogen bonding where as HCl is a gas due to there is no such type of bonding in it.
![]()
Question 11.
Bond dissociation enthalpy of F2 is less than that of Cl2 – explain.
Ans. Bond dissociation enthalpy of F2 is less than that of Cl2
Explanation:
In F2 molecule electron repulsions are greater among lone pairs because these lone pairs are much closer to each other than in case of Cl2.
Question 12.
Write the formulae of the compounds, in which oxygen has positive oxidation states and mention the oxidation states of oxygen in them.
Answer:
- In OF2 and O2F2 oxygen has positive oxidation states.
- In OF2, oxygen oxidation state is + 2.
- In O2F2 oxygen oxidation state is + 1.
Question 13.
What is the use of O2F2 and I2O5?
Answer:
Use of O2F5:
O2F2 is a fluorinating agent. O2F2 oxidises plutonium to PUF6 and the reaction is used in removing plutonium as PUF6 from spent nuclear fuel.
Use of I2O5:
I2O5 is a good oxidising agent and is used in the estimation of carbon monoxide (CO).
Question 14.
Write two uses of hydrogen chloride.
Answer:
Uses of hydrogen chloride :
- It is used in medicines and as a laboratory reagent.
- It is used in the manufacturing of Cl2, NH4Cl and glucose (from com starch).
- It is used in extracting glue from bones-and purifying bone black.
![]()
Question 15.
Explain the reactions of Cl2 with NaOH. [T.S. Mar. 16]
Answer:
- Reaction with cold dilute NaOH: Chlorine reacts with cold dilute NaOH to give sodium hypochlorite and sodium chloride.
2 NaOH (cold, dil.) + Cl2 → NaCl + NaOCl + H2O - Reaction with hot concentrated NaOH : Chlorine reacts with hot concentrated NaOH to give sodium chlorate and sodium chloride.
3 Cl2 + 6 NaOH (hot, Conc.) → 5 NaCl + NaClO3 + 3 H2O
Question 16.
What happens when Cl2 reacts with dry slaked lime ? [A.P. Mar. 16, 15]
Answer:
Chlorine reacts with dry slaked lime and forms bleaching powder.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
Question 17.
Chlorine acts as an oxidizing agent – explain with two examples.
Answer:
Chlorine acts as oxidising agent.
Example – 1: Cl2 oxidises Iodine to Iodate .
I2 + 6 H2O + 5 Cl2 → 2HIO3 + 10 HCl
Example – 2 : Cl2 oxidises Sodium Sulphite to Sodium Sulphate
Cl2 + Na2SO3 + H2O → Na2SO4 + 2 HCl
Question 18.
What is aqua regia ? Write its reaction with gold and platinum.
Answer:
A mixture of 3 parts of Cone. HCl and one part of Cone. HNO3 constitutes aqua regia. It is used for dissolving noble metals.
It’s reaction with gold :
Au + 4H+ + [latex]\mathrm{NO}_3^{-}[/latex] + 4Cl– → [latex]\mathrm{AuCl}_4^{-}[/latex] + NO + 2 H2O
It’s reaction with Platinum
3Pt + 16H+ + 4[latex]\mathrm{NO}_3^{-}[/latex] + 18 Cl– → [latex]3 \mathrm{PtCl}_6^{-2}[/latex] + 4 NO + 8H2O
Question 19.
How is chlorine manufactured by Deacon’s method ? [T.S. & A.P. Mar. 19 & T.S. Mar. 16]
Answer:
Deacon’s process : In Deacon’s process chlorine is obtained by the oxidation of hydrogen chloride gas by atmospheric oxygen in the presence of CuCl2 (catalyst) at 723 K.
![]()
![]()
Question 20.
Chlorine acts as a bleaching agent only in the presence of moisture – explain.
Answer:
Moist chlorine is a powerful bleaching agent. This bleaching property is due to oxidation.
Cl2 + H2O → 2HCl + (O)
Ex : Coloured substance + (O) → colourless substance.
Question 21.
The decreasing order of acidic character among hypohalogen acids is HClO > HBrO >HIO. Give reason.
Answer:
Given the decreasing order of acidic character among hypohalogen acids is
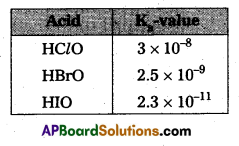
HaO > HBrO > HIO
From the above mentioned Ka values the given order of hypohalogen acids – acid strength order is
HClO > HBrO > HIO
Question 22.
The acidic nature of the oxoacids of chlorine is
HOCl < HClO2 < HClO3 < HClO4 – explain.
(Hint: HA + H2O ⇌ H3O+ + A– conjugate base, greater the stability of A–, lesser will be its basic strength or greater will be the tendency of HA to release H+. In other words, stronger will be the acid HA. Among the conjugate bases of oxoacids of chlorine, the stability order is HOCl– < [latex]\mathrm{ClO}_2^{-}[/latex] > [latex]\mathrm{ClO}_3^{-}[/latex] > [latex]\mathrm{ClO}_4^{-}[/latex])
Answer:
Given the acidic nature of oxoacids of chlorine is
HOCl– < H[latex]\mathrm{ClO}_2^{-}[/latex] > H[latex]\mathrm{ClO}_3^{-}[/latex] > [latex]\mathrm{ClO}_4^{-}[/latex]
HA + H2O ⇌ H3O+ + A– conjugate base, greater the stability of A–, lesser will be its basic strength or greater will be the tendency of HA to release H+. In other words, stronger will be the acid HA. Among the conjugate bases of oxoacids of chlorine, the stability order is
HOCl– < H[latex]\mathrm{ClO}_2^{-}[/latex] > H[latex]\mathrm{ClO}_3^{-}[/latex] > H[latex]\mathrm{ClO}_4^{-}[/latex]
![]()
Question 23.
What are interhalogen compounds ? Give two examples.
Answer:
The binary diamagnetic compounds of halogens which are formed by the reaction of halogens among themselves are called interhalogen compounds.
E.g.: IF7, ClF3, BrF3, ClF, IF3 etc.
Question 24.
Explain the structure of ClF3.
Answer:
Structure of ClF3:
- Central atom in ClF3 is ‘Cl’
- Excited state electronic configuration of ‘Cl’ is
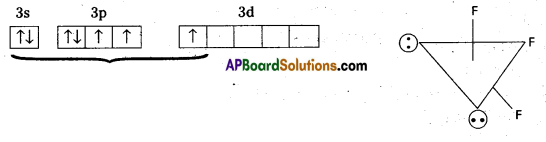
- Cl atom undergoes sp3d hybridisation
- It is a bent T-shaped molecule (or) trigonal bipyramidal with 2 – positions occupied by lone pairs.
Question 25.
OF2 should be called oxygen difluolide and not fluorine oxide – Why ?
Answer:
OF2 should be called oxygen difluoride and riot fluoride oxide.
The binary compotmds of oxygen with fluoride are not pronounced as oxides because the electronegativity of fluorine is greater than oxygen.
Question 26.
Iodine is more soluble in KI than in water – Explain.
Answer:
Iodine is more soluble in KI than in water.
Reason:
Iodine combine with KI forming soluble complex KI3.
KI + I2 → KI3 (Soluble complex)
Iodine does not dissolve in water. This is due to positive free energy change (+ ∆G)
![]()
Question 27.
Among the hydrides of halogens
a) Which is most stable ?
b) Which is most acidic ?
c) Which has lowest boiling point ?
Answer:
a) Among hydrides of halogens HF is most stable.
b) Among hydrides of halogens HI is most acidic.
c) Among hydrides of halogens HCl (189K) has lowest boiling point.
Question 28.
Compare the bleaching action of Cl2 and SO2.
Answer:
- Cl2 is a powerful bleaching agent. This bleaching action is due to oxidation.
Cl2 + H2O → 2HCl+ (O)
Coloured substance + (O) → colourless substance. - It bleaches vegetable or organic matter in the presence of moisture. Bleaching effect of Cl2 is permanent.
- SO2 acts as bleaching agent in presence of moisture.
SO2 + 2 H2O → H2SO4 + 2[H]
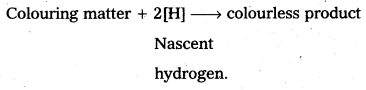
- SO2 bleaches wool and silk.
Question 29.
Give the oxidation states of halogens in the following :
a) Cl2O
b) [latex]\mathrm{ClO}_2^{-}[/latex]
c) KBrO3
d) NaClO4
Answer:
a) Cl2O :
2x – 2 = 0
x = -+ 1
Oxidation state of chlorine in Cl2O is + 1.
b) [latex]\mathrm{ClO}_2^{-}[/latex]
x + 2(-2) = -1
x = -1 + 4 = +3
In latex]\mathrm{ClO}_2^{-}[/latex] the oxidation state of ‘Cl’ is + 3
c) KBrO3 :
1 + x + 3 (-2) = 0
x = + 5
In KBrO3 the oxidation state of Br is + 5.
d) NaClO4
1 + x + 4(-2) = 0
x = + 7
In NaClO4 the oxidation state of Cl is + 7.
![]()
Question 30.
Describe the molecular shape of [latex]\mathrm{I}_3^{-}[/latex] .
(Hint: Central iodine is of sp3d. – linear)
Answer:
- In Tri iodide ion central iodine atom undergo sp3d hybridisation.
- It contains three lone pairs and two bond pairs.
- According to VSEPR theory it has linear shape.

Short Answer Questions
Question 1.
How can you prepare Cl2 from HCl and HCl from Cl2 ? Write the reactions.
Answer:
Preparation of Cl2 from HCl:
- On heating MnO2 with Conc. HCl, Cl2 gas is liberated
MnO2 + 4HCl → MnCl2 + Cl2 + 2H2O - By the Õxidation of HCl gas by atmospheric oxygen in presence of CuCl2 catalyst at 723 K.
4 HCl + O2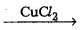 2Cl2 + 2 H2O
2Cl2 + 2 H2O
Preparation of HCl from Cl2:
- Cl2 when reacted with H2 to form HCl
H2(g) + Cl2(g) → 2HCl(g)
Question 2.
Write balanced equations for the following.
a) NaCl is heated with Conc.H2SO4 In the presence of MnO2.
b) Chlorine is passed into a solution of Nal ¡n water.
Answer:
a) NaCl is heated with Conc.H2SO4 in presence of MnO2 liberates Cl2 gas.
4 NaCl + MnO2 + 4 H2SO4 → MnCl2 + 4 NaHSO4 + 2 H2O + Cl2.
b) When chlorine water is added to a solution of sodium iodide a brown colour is formed.
Cl2 + 2 NaI → 2 NaCl + I2
Question 3.
Explain the structures of
a) BrF5 and
b) IF2.
Answer:
a) Structure of BrF5.
- Central atom in BrF5 is Br.
- ‘Br’ undergoes sp3d2 hybridisation in 2nd excited state.
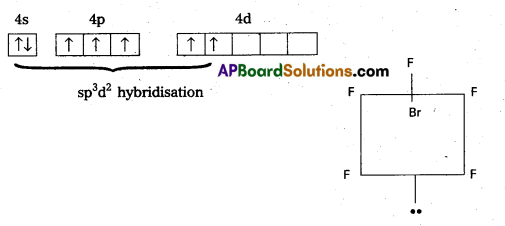
- Shape of the molecule is octahedral with one position occupied by a lone pair (or) square pyramidal.
![]()
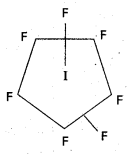
b) Structure of IF7 :
- Central atom in IF7 is T.
- T undergoes sp3d3 hybridisation in 3rd excited state.
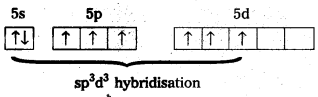
- Shape of the molecule is Penta gonal bipyramid structure
Question 4.
Write a short note on the hydrides of halogens.
Answer:
Hydrides of halogens :
Formation :
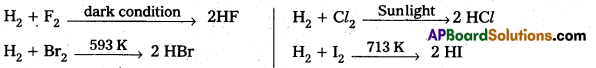
These compounds dissolve in water to form hydrohalic acids.
Boiling points HF – 293 K
HCl – 189K
HBr – 206 K
HI – 238 K
The decreasing order of stability of these compounds is
HF > HCl > HBr > HI
The increasing order of acidic strength of these compounds
HF < HCl < HBr < HI
Question 5.
How is chlorine obtained in the laboratory ? How does it react with the following ? [T.S. Mar. 15]
a) cold dil. NaOH
b) excess NH3
c) KI
Answer:
In the laboratory chlorine is prepared by the oxidation of HCl with MnO2.
4 HCl + MnO2 → MnCl2 + 2 H2O + Cl2 ↑
a) Chlorine reacts with cold dil. NaOH to form sodium hypochlorite.
Cl2 + 2 NaOH → NaCl + NaOCl + H2O
b) Cl2 reacts with excess of NH3, Nitrogen and ammonium chloride are formed.
8 NH3 + 3 Cl2 → 6 NH4Cl + N2 ↑
c) Cl2 reacts with KI to liberate iodine
Cl2 + 2KI → 2 KCl + I2 ↑
![]()
Question 6.
What are interhalogen compounds ? Give some examples to illustrate the definition. How are they classified ?
Answer:
The binary diamagnetic compounds of halogens which are formed by the reaction of halogens among themselves are called interhalogen compounds.
E.g.: IF7, ClF3, BrF3, ClF, IF3 etc.
The above examples are binary diamagnetic compounds and formed by* combination of halogens only. .
Inter halogen compounds are classified into four types.
- AX – Type : Eg : ClF, BrF
- AX3 – Type : Eg: ClF3, IF3
- AX5 – Type : Eg : ClF5, BrF5
- AX7 – Type : Eg : IF7
‘A’ is less electronegative halogen.
X is more electronegative halogens.
Long Answer Questions
Question 1.
How is ClF3 prepared ? How does it react with water ? Explain its structure.
Answer:
Preparation of ClF3: Chlorine reacts with excess of fluorine to form ClF3.
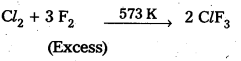
Reaction with H2O: ClF3 reacts with water explosively and oxidises water to give oxygen or in controlled quantities oxygen diflouride (OF2) as well as HF and HCl.
ClF3 + 2H2O → 3 HF + HCl + O2
ClF3 + H2O → 4 HF + HCl + OF2
Structure of ClF3:
- Central atom in ClF3 is ‘Cl’
- Excited state electronic configuration of ‘Cl’ is
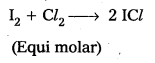
- Cl atom undergoes sp3d hybridisation
- It is a bent T-shaped molecule (or) trigonal bipyramidal with 2 – positions occupied by lone pairs.
![]()
Question 2.
How is chlorine prepared in the laboratory ? How does it react with the following ? [T.S. Mar. 19, 15]
a) Iron
b) hot, cone. NaOH
c) acidified FeSO4
d) Iodine
e) H2S
f) Na2S2O3
Answer:
In the laboratory chlorine is prepared by the oxidation of HCl with MnO2.
4 HCl + MnO2 → MnCl2 + 2 H2O + Cl2 ↑
a) Cl2 reacts with Iron to form FeCl3
2 Fe + 3Cl2 → 2 FeCl3
b) Cl2 reacts with hot Cone. NaOH to form sodium chloride and sodium chlorate
3Cl2 + 6 NaOH → 5 NaCl + NaClO3 + 3 H2O
c) Cl2 reacts with acidified FeSO4 and ferric ions are formed.
2 FeSO4 + H2SO4 + Cl2 → Fe2(SO4)3 + 2 HCl
d) Cl2 reacts with Iodine to form ICl
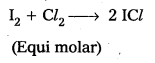
e) Cl2 reacts with H2S and forms HCl and ‘S’
Cl2 + H2S → 2 HCl + S
f) ‘S’ is precipitated by the reaction of Cl2 with Na2S2O3.
‘Na2S2O3 + Cl2 + H2O → Na2SO4 + 2 HCl + S
![]()
Question 3.
Discuss the anomalous behaviour of fluorine.
Answer:
Abnormal behaviour of fluorine : Fluorine being the first element in the group differs considerably from other halogens. The reasons may be traced out to one or more of the following, characteristics.
F2 has
- a small size
- The highest electronegativity amongst the known elements.
- No d-orbital are available in its valence shell.
- Low enthalpy of F-F bond dissociation and
- Only two electrons are present in the penultimate shell while other halogens 8 electrons.
The abnormal characteristics of F2 can be summarised as follows.
- F2 exhibits -1 oxidation state because it is the most electronegative element known. Therefore, no higher oxidation state for it in its compounds.
- In it’s hydride HF, it has hydrogen bonding, but other halides of hydrogen do not show this property. HF can form [latex]\mathrm{HF}_2^{-}[/latex] ion. No such ions are known with other halogens.
- It combines with carbon while others do not combine even under drastic conditions.
- F2 has a lower EA compared to that of Cl2 even though F2 is the most electronegative element.
- Fluorides have the highest ionic character among the halides.
Example : AlF2 is ionic while AlCl3 is covalent compound.
Question 4.
How is chlorine prepared by electrolytic method ? Explain its reaction with [A.P. Mar. 16, 15]
a) NaOH and
b) NH3 under different conditions.
Answer:
Preparation of chlorine by electrolytic method : Chlorine is obtained by the electrolysis of brine solutions (Cone. NaCl). Cl2 gas is liberated at anode.
2 NaCl → 2 Na+ + 2Cl–
2 H2O + 2e– → 2 OH– + H2 (cathode)
2 Cl– → Cl2 + 2e– (anode)
a)
- NaOH:
Chlorine reacts with cold dil. NaOH to form sodium hypochlorite.
Cl2 + 2 NaOH → NaCl + NaOCl + H2O - Cl2 reacts with hot cone. NaOH to form sodium chloride and sodium chlorate.
3 Cl2 + 6 NaOH → 5 NaCl + NaClO3 + 3 H2O
b)
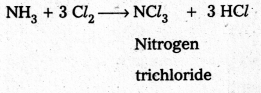
- Cl2 reacts with excess of NH3, Nitrogen and ammonium chlorate are formed.
8 NH3 + 3 Cl2 → 6 NH4Cl + N2 ↑ - NH3 reacts with excess of Cl2 to form NCl3 and HCl.
![]()
Question 5.
Write the names and formulae of the oxoacids of chlorine. Explain their structures and relative acidic nature.
Answer:
Four oxyacids of chlorine are known. They are
Hypochlorous acid – HOCl
Chlorous acid – HClO2
Chloric acid – HClO3
Perchloric acid – HClO4
Structure of HClO : In this chlorine atom is sp3 hybridised. Outer electronic configuration of Cl in ClO– after sp3 hybridisation.

Shape is tetrahedral with 3 lone pairs (or) linear.
No π – bonds.
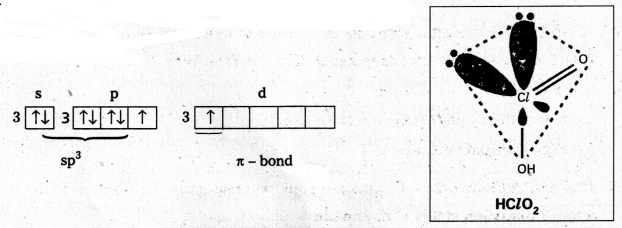
Chlorous acid : (HClO2) : Chlorine is in sp3 hybrid state, in first excited state. Shape is tetrahedral with 2 lone pairs (or) angular one πd-p bond is present.
First excited state
Chloric acid (HClO3) : The central chlorine atom undergoes sp3 hybridisation in second excited state.
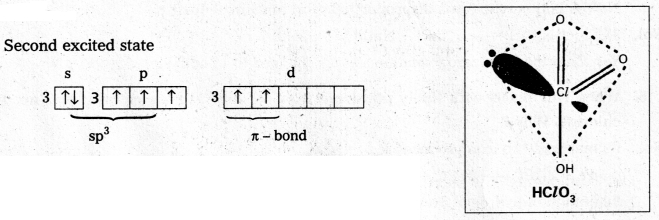
Shape is tetrahedral with one lone pair (or) pyramidal.
Two πd-p bonds present.
Perchloric acid (HClO4) : The central chlorine atom undergoes sp3 hybridisation in third excited state.
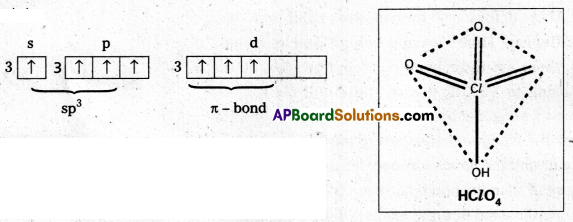
Shape is perfect tetrahedral. No lone pairs.
Three πd-p bonds present.
Textual Examples
Question 1.
Halogens have maximum negative electron gain enthalpy in the respective periods of the periodic table. Why ?
Solution:
Halogens have the smallest size in their respective periods and therefore high effective nuclear charge. As a consequence, they readily accept one electron to acquire noble gas electronic configuration.
Question 2.
Although electron gain enthalpy of fluorine is less negative as compared to chlorine, fluorine is a stronger oxidising agent than chlorine. Why ?
Solution:
It is due to
- Low enthalpy of dissociation of F-F bond
- High hydration enthalpy of F–
![]()
Question 3.
Fluorine exhibits only – 1 oxidation state whereas other halogens exhibit +1, +3, +5 and +7 oxidation states also. Explain.
Solution:
Fluorine is the most electronegative element and cannot exhibit any positive oxidation .state. Other halogens have d orbitals and therefore, can expand their octets and show +1, +3, +5 and +7 oxidation states also.
Question 4.
Write the balanced chemical equation for the reaction of Cl2 with hot and concentrated NaOH. Is this reaction a disproportionation reaction ? Justify.
Solution:
3 Cl2 + 6 NaOH → 5NaCl + NaClO3 + 3H2O
Yes. Chlorine from zero oxidation state is changed to -1 and +5 oxidation states.
Question 5.
When HCl reacts with finely powdered iron, it forms ferrous chloride and not ferric chloride. Why ?
Solution:
Its reaction with iron produces H2.
Fe + 2HCl → FeC2 + H2
Liberation of hydrogen prevents the formation of ferric chloride.
![]()
Question 6.
Discuss the molecular shape of BrF3 on the basis of VSEPR theory.
Solution:
The central atom Br has seven electrons in the valence shell. Three of these will form electron pair bonds with three fluorine atoms leaving behind four electrons. Thus, there are three bond pairs and two lone pairs.
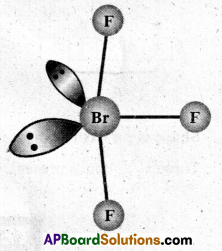
According to VSEPR theory, these will occupy the comers of a trigonal bipyramid. The two lone pairs will occupy the equatorial positions to minimise lone pair- lone pair and the bond pair-lone pair repulsions which are greater than the bond pair-bond pair repulsions. In addition, the axial flourine atoms will be bent towards the equatorial fluorine in order to minimise the lone pair-lone pair repulsions. The shape would be that of a slightly bent T.
Intext Questions
Question 1.
Considering the parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy, compare the oxidising power of F2 and Cl2.
Oxidizing power is a combined effect of bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
Solution:
Comparing F2 and Cl2 with the given parameters.

From the data given above, it is clear that the bond dissociation enthalpy and electron gain enthalpy are higher for chlorine but hydration energy is much higher for fluorine. It compensates the effect of other two and thus, makes flourine more oxidizing than chlorine.
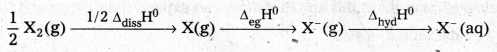
The relative oxidizing power of the halogens can be further illustrated by their reactions with water.
2 F2(g) + 2 H2O(l) → 4H+ (aq) + 4F– (aq) + O2(g) ……………. (i)
Cl2(g) + H2O(l) → HCl (aq) + HOCl (aq) ……………… (ii)
Question 2.
Give two examples to show the anomalous behaviour of fluorine.
Solution:
The two anomalous behaviour of flourine are as follows :
a) It forms only one oxo-acid while other halogens form a number of oxo-acids.
b) Hydrogen flouride (HF) is a liquid (b.p. 293 K) due to strong hydrogen bonding whereas other hydrogen halides are gases.
Question 3.
Sea is the greatest source of some halogens. Comment.
Solution:
Sea water contains chlorides, bromides and iodides of sodium, potassium, magnesium and calcium. NaCl is the most abundant (2.5% by mass) among all of these. Certain forms of marine life contain iodine in their systems; Various sea weeds, for example, contain upto . 0.5% of iodine and chile saltpetre contains upto 0.2% of sodium iodate. NaCl and camalite, KCl, MgCl2. 6H2O etc are present in the deposits of dried up seas.
![]()
Question 4.
Give the reason for bleaching action of Cl2 ?
Solution:
Bleaching action of chlorine is due to its oxidizing property. When chlorine reacts with water, it gives nascent oxygen which decoloures is the coloured substance.
Cl2 + H2O → 2HCl + [O]
Coloured substance + [O] → Colourless substance
Bleaching action of chlorine creates permanent effect. It bleaches the vegetable or organic matter in the presence of moisture.
Question 5.
Name some poisonous gases which can be prepared from chlorine gas.
Solution:
- Phosgene (COCl2)
- Tear gas (CCl3NO2)
- Mustard gas (ClCH2CH2SCH2CH2Cl)