Practice the AP 10th Class Physical Science Bits with Answers Chapter 12 Carbon and its Compounds on a regular basis so that you can attempt exams with utmost confidence.
AP State Syllabus 10th Class Physical Science Bits 12th Lesson Carbon and its Compounds with Answers
Concept – 1 : Versatile Nature of Carbon
Question 1.
The following is not correct about carbon
A) It is a non metal
B) It belongs to 14th group
C) Its electron configuration is 1s2 2s22p2
D) Its valency is 3
Answer:
D) Its valency is 3
![]()
Question 2.
The electronegativity of carbon is
A) 1.5
B) 2.5
C) 3.5
D) 4
Answer:
B) 2.5
Question 3.
Thecovalency of carbon is
A) 1
B) 2
C) 3
D) 4
Answer:
D) 4
Question 4.
Match the following:
a) Methane — i) sp
b) Ethylene or ethene — ii) sp3
c) Acetylene or ethyne — iii) sp2
A) a → i, b → ii, c → iii
B) a → iii, b → ii, c → i
C) a → ii, b → iii, c → i .
D) a → ii, b → i, c → iii
Answer:
C) a → ii, b → iii, c → i
Question 5.
Match the following:
a) Methane — i), 180°
b) Ethene — ii) 109° 28′
c) Ethyne — iii) 120°
A) a → i, b → ii, c → iii
B) a → iii, b → ii, c i
C) a → ii, b → i, c → iii
D) a → ii, b → iii, c → i
Answer:
D) a → ii, b → iii, c → i
Question 6.
Carbon (Z=6) is the element of group-IVA of periodic table. Which of the following is the atomic number of next number in the group
A) 9
B) 14
C) 15
D) 17
Answer:
B) 14
Question 7.
The number of sigma and pi bonds in ethene are …………,………………respectively.
A) 1, 5
B) 2, 3
C) 3,2
D) 5,1
Answer:
D) 5,1
![]()
Question 8.
Number of sigma and pi bonds in ethyne are ……………
A) 2, 3
B) 3, 2
C) 4,1
D) 1, 4
Answer:
B) 3, 2
Question 9.
Ethylene is common name for ……………….
A) ethene
B) ethane
C) ethyne
D) ethanol
Answer:
A) ethene
Question 10.
Acetylene is common name for ……………..
A) ethane
B) ethene
C) ethyne
D) ethanol
Answer:
C) ethyne
Question 11.
The number of a bonds in methane is …………………
A) 2
B) 3
C) 4
D) 1
Answer:
C) 4
Question 12.
The electronic configuration of carbon in excited state is ………………….
A) 1s2 2s2 2p1y2p1z
B) 1s2 2s1 2p1x2p1y2p1z
C) 1s2 2s2 2p1x 2p1y
D) 1s2 2s2 2p2x
Answer:
B) 1s2 2s1 2p1x2p1y2p1z
Question 13.
The bond angle in C2H2 molecule is
A) 109° 28′
B) 120°
C) 180°
D) 107°
Answer:
D) 107°
Question 14.
How many unpaired electrons the carbon atom posseses in the ground state?
A) 2
B) 3
C) 4
D) 1
Answer:
A) 2
Question 15.
The type of hybridization in CH4 molecule is ………..
A) sp
B) sp2
C) sp3
D) sp3d
Answer:
C) sp3
Question 16.
Which of the following molecules undergo sp hybridisation ?
A) CH4
B) C2H4
C) C2H6
D) C2H2
Answer:
D) C2H2
Question 17.
Who among the following disproved vitalism theory ?
A) Berzelius
B) Linus pauling
C) Autbau
D) Wohler
Answer:
D) Wohler
Question 18.
The bond angle in C2H4 is
A) 109° 28′
B) 180°
C) 104° 27′
D) 120°
Answer:
D) 120°
![]()
Question 19.
The chemical bonds present in C2H2 are
A) 2 sigma and 3 pi
B) 1 sigma and 2 pi
C) 3 sigma and 2 pi
D) 2 sigma and 1 pi
Answer:
C) 3 sigma and 2 pi
Question 20.
n – butane and isobutane are
A) structural isomers
B) functional isomers
C) stereo isomers
D) geometrical isomers
Answer:
A) structural isomers
CONCEPT – II – Allotropes of Carbon
Question 21.
The property of an element to exist in two or more physical forms with same chemical properties but different physical properties is called
A) catenation
B) isomerism
C) allotropy
D) conjugation
Answer:
C) allotropy
Question 22.
The amorphous forms of carbon allutropes
i) coal
ii) coke
iii) animal charcoal
iv) diamond
A) only i
B) only ii
C) both i and ii
D) i, ii and iii
Answer:
D) i, ii and iii
Question 23.
The crystalline forms of carbon allotropes
i) diamond
ii) graphite
iii) lamp black
iv) buckminster fullerene
A) only i
B) both i and ii
C) i, ii and iv
D) all of these
Answer:
C) i, ii and iv
Question 24:
The hybridisation present in diamond is
A) sp
B) sp2
C) sp3
D) dsp2
Answer:
C) sp3
Question 25.
The hardest material among these is ……………….
A) diamond
B) graphite
C) buckminster fullerene
D) coke
Answer:
A) diamond
Question 26.
In diamond each carbon atom has a …………………… environment.
A) trigonal planar
B) tetrahedral
C) square planar
D) pyramidal
Answer:
B) tetrahedral
Question 27.
Graphite forms ……………. layer structure.
A) one dimensional
B) two dimensional
C) three dimensional
D) none of these
Answer:
B) two dimensional
Question 28.
In layer structure of graphite the carbon atoms are in ………………. environment.
A) trigonal planar
B) tetrahedral
C) square planar
D) pyramidal
Answer:
A) trigonal planar
Question 29.
The hybridisation present in graphite is …………………….
A) sp
B) sp2
C) sp3
D) d2sp3
Answer:
B) sp2
Question 30.
The distance between two successive layers of graphite is…………….
A) 1.35 A°
B) 2.35 A°
C) 3.35 A°
D) 4.35 A0
Answer:
C) 3.35 A°
![]()
Question 31.
The use of graphite is ……………….
A) lubricant
B) lead in pencils
C) good conductor of electricity
D) all of these
Answer:
D) all of these
Question 32.
Delocalized n system is present in …….
A) diamond
B) charcoal
C) coal
D) graphite
Answer:
D) graphite
Question 33.
Graphite is a good conductor of electricity due to ………………….
A) delocalized a system
B) localized n system
C) delocalized n system
D) localized a system
Answer:
C) delocalized n system
Question 34.
Buckminster fullerene was discovered by ………………
A) Robert F. Curl
B) Harold W.Kroto
C) Richard E. Smalley
D) All of these
Answer:
D) All of these
Question 35.
Bucky balls are ………………. fullerences.
A) cylindrical
B) spherical
C) conical
D) elliptical
Answer:
B) spherical
Question 36.
The shape of Buckminster fullerene is ………………..
A) soccer ball
B) cricket ball
C) basket ball
D) base ball
Answer:
A) soccer ball
Question 37.
The pentagonal and hexagonal faces in Buckminster fullerene are ……………… and ………………. respectively.
A) 12, 20
B) 20,12
C) 12,18
D) 18,12
Answer:
A) 12, 20
Question 38.
The hybridisation present in Buckminster fullerene is ……………….
A) sp3
B) sp2
C) sp
D) sp3d2
Answer:
B) sp2
Question 39.
……. is useful in curing melanoma cancer.
A) Diamond
B) Graphite
C) Fullerene
D) Nano tube
Answer:
C) Fullerene
Question 40.
Nano tubes were discovered by …………………
A) H. W. Kroto
B)-RE. Smalley
C) Sumi Li Jima
D) R. F. Curl
Answer:
C) Sumi Li Jima
Question 41.
The uses of nano tubes are
i) used as molecular wires
ii) used in integrated circuits instead of copper
iii) biomolecules are inserted into nano tubes to inject them into a single cell
A) only i
B) both i and ii
C) i, ii and iii
D) none of these
Answer:
C) i, ii and iii
Question 42.
Graphene is extracted from
A) graphite
B) diamond
C) nano tube
D) coal
Answer:
A) graphite
Question 43.
…………. conducts electricity better than copper
A) Graphite
B) Graphene
C) Nano tubes
D) Buckminster fullerene
Answer:
B) Graphene
Question 44.
…………… named the compounds derived from living organisms as organic compound
A) Wohler
B) Berzelius
C) Lewis
D) Pauling
Answer:
B) Berzelius
Question 45.
The first synthetic carbon compound is …….. and it was prepared by …………
A) Acetic acid, Wohler
B) Alcohol, Berzelius
C) Urea, Wohler
D) Dimethyl, Lewis
Answer:
C) Urea, Wohler
![]()
Question 46.
The formula of Urea is
A) NH4CNO
B) CO(NH2)2
C) CH3CONH2
D) C3H7NH2
Answer:
B) CO(NH2)2
Question 47.
The formula of ammonium cyanate is ……………..
A) NH4CNO
B) CO(NH2)2
C) CH3COONa
D) CH3NH2
Answer:
A) NH4CNO
Question 48.
The ability to form longest chains with its own atoms is ………..
A) Allotropy
B) Isomerism
C) Catenation
D) Combustion
Answer:
C) Catenation
Question 49.
The following elements show catenation.
A) carbon
B) sulphur
C) phosphorous
D) all of these
Answer:
D) all of these
Question 50.
The properties which made carbon as versatile element
i) Tetra valency
ii) Catenation
iii) Isomerism
iv) Formation of multiple bonds
A) only i
B) only ii
C) both i and ii
D) All of these
Answer:
D) All of these
Question 51.
The hardest material among the allotropes of carbon is
A) diamond
B) graphite
C) coke
D) coal
Answer:
A) diamond
Question 52.
Which of the following is not an electric conductor?
A) nano tube
B) graphene
C) diamond
D) graphite
Answer:
C) diamond
Question 53.
In integrated circuit ………….. are used instead of copper to connect the components together
A) graphite
B) C60
C) nano tubes
D) PVC
Answer:
C) nano tubes
CONCEPT – III : Hydro Carbons
Question 54.
Hydrocarbons contains the following elements
A) H, C
B) H, CO
C) N, C
D) H, Cl
Answer:
A) H, C
Question 55.
The characteristic properties of an organic compound which depend on atom or group of atoms in its molecule is known as …………………
A) standard group
B) functional group
C) associate group
D) alkyl group
Answer:
B) functional group
Question 56.
The phenomenon of possessing same molecular formula but different properties by compounds in known as ……..
A) Allotropy
B) Catenation
C) Isomerism
D) Combustion
Answer:
C) Isomerism
Question 57.
Which of the following is a saturated hydrocarbon?
A) C2H4
B) C2H2
C) C3H6
D) C2H6
Answer:
D) C2H6
Question 58.
The general formula of alkynes is
A) C2H2n+2
B) CnH2n-2
C) CnH2n
D) CnH2n+1
Answer:
B) CnH2n-2
Question 59.
Match the following :
a) Alkanes — i) Hydrocarbons containing double bonds
b) Alkenes — ii) Hydrocarbons containing triple bonds
c) Alkynes — iii) Hydrocarbons containing single bonds
A) a → i, b → iii, c → ii
B) a → i, b → ii, c → iii
C) a → iii, b → i, c → ii
D) a → ii, b → iii, c → i
Answer:
C) a → iii, b → i, c → ii
Question 60.
Match the following :
a) Alkanes i) C2H2n-2
b) Alkenes ii) C2H2n+2
c) Alkynes iii) C2H2n
A) a → ii, b → iii, c → i
B) a → i, b → ii, c → iii
C) a → iii, b → ii, c → i
D) a → iii, b → i, c → ii
Answer:
a) Alkanes i) C2H2n-2
Question 61.
The hydrocarbon having single bonds between the carbon atoms is ……………
A) Ethane
B) Ethene
C) Propene
D) Butyne
Answer:
A) Ethane
![]()
Question 62.
The number of hydrogen atoms in octane is ………………
A) 8
B) 18
C) 16
D) 14
Answer:
B) 18
Question 63.
To detect the leakage of gas from the cylinder the substance added to the gas is……..
A) C2H5SH
B) C2H5OH
C) CH3COOH
D) CH3CHO
Answer:
A) C2H5SH
Question 64.
Which of the following compound is not a hydrocarbon ?
A) R—CH3
B) RCH —CH2
C) RCH2OH
D) CH3CH = CH2
Answer:
B) RCH —CH2
CONCEPT – 4 : Nomenclature of Hydrocarbons
Question 65.
IUPAC name of alkene containing 3 carbon atoms is ………………..
A) ethene
B) propene
C) butene
D) pentene
Answer:
B) propene
Question 66.
Carbon compounds containing double bonds and triple bonds are called ………….. hydrocarbons.
A) saturated
B) unsaturated
C) super saturated
D) none of these
Answer:
B) unsaturated
Question 67.
Number of single covalent bonds in ammonia is . …………..
A) 1
B) 2
C) 3
D) 4
Answer:
C) 3
Question 68.
Which one of the following hydrocarbon can show isomerism
A) C2H4
B) C2H6
C) C3H8
D) C4H10
Answer:
D) C4H10
Question 69.
The number of structural isomers for pentane is
A) 2
B) 3
C) 4
D) 5
Answer:
B) 3
Question 70.
The series of carbon compounds in which two successive compounds differ -CH2 unit is called …………..
A) Reactivity series
B) Homologous series
C) Lyman series
D) Balmer series
Answer:
B) Homologous series
Question 71.
Match the following:
a) Alcohol — i) — CHO
b) Aldehyde — ii) OH
c) Ketone — iii) COOH
d) Acid — iv) C0
A) a → i1b →ii,c →iii,d →iv
B) a →iv,b →iii,c →ii,d →i
C) a →ii,b →i,c →iv,d →iii
D)a →iv,b →ii,c →iii,d →i
Answer:
C) a →ii,b →i,c →iv,d →iii
Question 72.
Match the following
a) Butanol — i) CH3-CH2-CH2-CH2-COOH
b) Pentanoic acid — ii) CH3 – CH2 – CH2 – CH2 – CH2– CHO
c) Propanone — iii) CH3-CH2-CH2-CH2-OH
d) Hexanal — iv) CH3COCH3
A) a → i,b → ii,c → iii,d → iv
B) a → iv,b → iii,c → ii,d → i
C) a → iii,b → i,c → iv,d → ii
D) a → iv,b → ii,c → iii,d → i
Answer:
C) a → iii,b → i,c → iv,d → ii
Question 73.
A compound which is a basic constituent of many cough syrups
A) Methanol
B) Ethanol
C) Ethyl alcohol
D) Both B and C .
Answer:
D) Both B and C .
Question 74.
Very dilute solution of ethanoic acid is …………..
A) Vinegar
B) Baking soda
C) Washing soda
D) Quick lime
Answer:
A) Vinegar
Question 75.
The functional group present in methanol is ……….
A) Alcohol
B) Aldehyde
C) Acid
D) Ketone
Answer:
A) Alcohol
![]()
Question 76.
The first member of homologous series among alkynes is ………………..
A) Methyne
B) Ethyne
C) Propyne
D) Butyne
Answer:
B) Ethyne
Question 77.
The suffix used for naming aldehyde is ………….
A) — ol
B) — al
C) — one
D) — ene
Answer:
B) — al
Question 78.
—NH2 group is called …………….
A) ester
B) ether
C) amine
D) amide
Answer:
C) amine
Question 79.
Combustion of hydrocarbon is generally accompanied by the evolution of ……………..
A) heat
B) light
C) both heat and light
D) electric current
Answer:
C) both heat and light
Question 80.
Oxidation of ethyl alcohol forms
A) ethanoic acid
B) acetic acid
C) formic acid
D) both A and B
Answer:
D) both A and B
Question 81.
The oxidizing agent used for conversion of alcohols to carboxylic acids is …………….
A) Alkaline potassium dichromate
B) Manganese dioxide
C) Acidified potassium dichromate
D) Both A and C
Answer:
D) Both A and C
Question 82.
The catalyst used in hydrogenation of oils is ………………..
A) Pd
B) Pt
C) Ni
D) Co
Answer:
C) Ni
Question 83.
Unsaturated organic compounds participate in …………….. reactions.
A) substitution
B) addition
C) elimination
D) rearrangement
Answer:
B) addition
Question 84.
A reaction in which an atom or a group of atoms in a compound is replaced by other atom or group of atoms is called
……………… reaction.
A) addition
B) elimination
C) substitution
D) rearrangement
Answer:
C) substitution
Question 85.
Paraffins are ………………..
A) Alkanes
B) Alkenes
C) Alkynes
D) Alcohols
Answer:
A) Alkanes
Question 86.
Saturated hydrocarbons (Alkanes) participate in ……………… reactions.
A) addition
B) substitution
C) elimination
D) rearrangement
Answer:
B) substitution
Question 87.
The process of conversion of starches and sugars to ethyl alcohol is called ……………..
A) carbonisation
B) esterification
C) condensation
D) fermentation
Answer:
D) fermentation
Question 88.
2 – Methyl propane is also called
A) Iso propane
B) n-butane
C) n – propane
D) Isobutane
Answer:
D) Isobutane
Question 89.
The IUPAC name of
HO – CH2– CH2– CH2– COOH.
A) 1 – hydroxy – 4 -butanoic acid
B) 4 – carboxyl – 1 – butanol
C) 3 – hydroxy -1- propanoic acid
D) 4 – hydroxy -1 – butanoic acid
Answer:
D) 4 – hydroxy -1 – butanoic acid
Question 90.
Esters are dervivatives of ………………….
A) Ketones
B) Alcohols
C) Ethers
D) Carboxylic acids
Answer:
D) Carboxylic acids
Question 91.
The name of the structure
CH3 – CH = C = CH2 is
A) Butene
B) Buta – 1, 2 – diene
C) Butadiene
D) 1, 3 – butadiene
Answer:
B) Buta – 1, 2 – diene
Question 92.
CH3 – NH – CH3 is known as
A) primary amine
B) teritituted amine
C) secondary amine
D) quaternary ammonium salt
Answer:
C) secondary amine
![]()
Question 93.
The general formula of ester is
A) R – O – R
B) R – CO – R
C) R-COOR
D) R – CHO
Answer:
C) R-COOR
Question 94.
The functional group in ketone is
A) 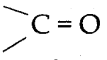
B) —COOH
C) —OH
D) — COOR
Answer:
A) 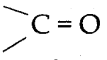
Question 95.
The functional group which is recognised as alcohol is
A) —CHO
B) —OH
C) —CO
D) —COOH
Answer:
B) —OH
Question 96.
The unsaturated hydrocarbon that contains multiple bonds undergo the following type of reaction to become a saturated hydrocarbon.
A) combustion
B) addition
C) oxidation
D) substitution
Answer:
B) addition
Question 97.
If CH3 – CH2 – CH2 – COOH is the IUPAC structure of carbon compound, then the carbon compound is
A) Butene
B) Butanol
C) Butanoic acid
D) But – 1 – ene
Answer:
C) Butanoic acid
Question 98.
If the carbon compound has many functional groups, then the order of preference while naming according to IUPAC nomenclature is
A) CONH2 > CHO > NH2 > — COOH
B) — COOH >CONH2 > CHO NH2
C) — CHO > CONH2 > COOH > NH2
D) — COOH > CHO > NH2 > CONH2
Answer:
B) — COOH >CONH2 > CHO NH2
Question 99.
The IUPAC name of
NH2 – CH2 – CHOH – CH2 – COOH is
A) 1 – amino – 2 hydroxy butanoic acid
B) 3 – hydroxy – 4 – amino butanoic acid
C) 4 – amino – 3 – hydroxy butanoic acid
D) 1 – amino – 3 – hydroxy butyric acid
Answer:
C) 4 – amino – 3 – hydroxy butanoic acid
Question 100.
IUPAC name of Cl3C – CH2 – CHO
A) 3, 3, 3 – trichloro propanal
B) 3, 3, 3 – trichloro propanol
C) 1,1,1 – trichloro propanol
D) 1,1,1 – trichloro propanal
Answer:
A) 3, 3, 3 – trichloro propanal
Question 101.
IUPAC name of
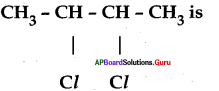
A) chloro butane
B) 2 – chloro butane
C) 2, 3 – chloro butane
D) 2, 3 – dichloro butane
Answer:
D) 2, 3 – dichloro butane
Question 102.
Simplest ketone is
A) CH3COCHg
B) CH3COCH2CH3
c) CH3COCH2CH2CH3
D) CH3CH2CHO
Answer:
A) CH3COCHg
Question 103.
Simplest acid is
A) Ethanoic acid
B) Methanoic acid
C) Propanoic acid
D) Butanoic acid
Answer:
B) Methanoic acid
Question 104.
The IUPAC name of following structure is
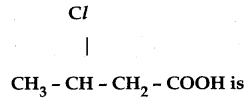
A) 2 – chloro – butanoic acid
B) 3 – chloro butanoic acid
C) 3 – chloro – pentanoic acid
D) 2 – chloro pentanoic acid
Answer:
B) 3 – chloro butanoic acid
CONCEPT – 5 : Some Important Carbon Compounds
Question 105.
Match the following:
a) Methyl chloride i) CHCl3
b) Methylene chloride ii) CH3Cl
c) Chloroform iii) CH2Cl2
d) Carbon tetra chloride iv) CCl4
A) a → i, b → ii, c → iii, d → iv
B) a → iv, b → iii, c → ii, d → i
C) a → iv, b → iii, c → i, d → ii
D) a → ii, b → iii, c → i, d → iv
Answer:
D) a → ii, b → iii, c → i, d → iv
Question 106.
Match the following:
a) Ethanol i) Acetic acid
b) Ethanoic acid ii) Acetaldehyde
c) Ethanal iii) Chloroform
d) Trichloro iv) Ethyl alcohol
methane
A) a → i, b → ii, c → iii, d → iv
B) a → iv, b → iii, c → ii, d → iv
C) a → iii, b → ii, c. → iv, d → i
D) a → iv, b → i, c → ii, d → iii
Answer:
D) a → iv, b → i, c → ii, d → iii
Question 107.
Pure ethanol is called …………….
A) absolute alcohol
B) rectified spirit
C) denaturated spirit
D) None of these
Answer:
A) absolute alcohol
Question 108.
Denaturated alcohol is ethanol that contains impurities like ……………..
A) Methanol
B) Methyl isobutyl ketone
C) Aviation gasoline
D) All of these
Answer:
D) All of these
Question 109.
When sodium metal is dropped in ethanol ………………. gas will be released.
A) Oxygen
B) Hydrogen
C) Chlorine
D) Nitrogen
Answer:
B) Hydrogen
Question 110.
The product that is formed by dehydration of ethanol in concentrated sulphuric acid is ……………….
A) Ethane
B) Ethene
C) Ethyne
D) Ethanal
Answer:
B) Ethene
Question 111.
Which of the four tubes containing
following chemicals show the brisk effervescence when dilute acetic acid is added to them ?
i) KOH
ii) NaHCO3
iii) K2CO3
iv) NaCl
A) i & ii
B) ii & iii
C) i&iv
D) i & iii
Answer:
B) ii & iii
Question 112.
A sweet odour substance formed by reaction of an alcohol and carboxylic acid is ……………………..
A) Ether
B) Aldehyde
C) Ketone
D) Ester
Answer:
D) Ester
Question 113.
Which of the following solution of acetic acid in water can be used as preservative.
A) 5 -10 %
B) 10 -15 %
C) 15 – 20 %
D) 100 %
Answer:
A) 5 -10 %
![]()
Question 114.
Acetic acid, when dissolved in water, dissociates into ions reversibly because it is a
A) weak acid
B) strong acid
C) weak base
D) strong base
Answer:
A) weak acid
Question 115.
2 ml of ethanoic acid was taken in three test tubes A, B and C and 2 ml, 4 ml and 8 ml water was added to them respectively. A clean solution is obtained in
A) Test tube A only
B) Test tubes A & B only
C) Test tubes B & C only
D) All the test tubes
Answer:
D) All the test tubes
Question 116.
If 2 ml of acetic acid is added slowly in drops to 5 ml of water then we will notice
A) the acid forms a separate layer on the top of water
B) water forms a separate layer on the top of the acid
C) formation of clear and homogeneous solution
D) formation of pink and clear solution
Answer:
C) formation of clear and homogeneous solution
Question 117.
A few drops of ethanoic acid were added to sodium carbonate. The possible results of the reaction :
A) A hissing sound was evolved
B) Brown fumes evolved
C) Brisk effervescence occurred
D) A pungent smelling gas evolved
Answer:
C) Brisk effervescence occurred
Question 118.
When acetic acid reacts with ethyl o alcohol, we add coiic.H2S04 . It acts as and the process is called
A) oxidizing agent, saponification
B) dehydrating agent, esterification
C) reducing agent, esterification
D) acid and esterification
Answer:
B) dehydrating agent, esterification
Question 119.
The strength of acids may be expressed in terms of
A) H+ ion concentration
B) pH
C) PKa
D) all of these
Answer:
D) all of these
Question 120.
Sodium or potassium salt of fatty acid is……
A) soap
B) detergent
C) indicator
D) soda
Answer:
A) soap
Question 121.
Match the following
a) Palmitric acid — i) C17H35COOH
b) Stearic acid — ii) C17H33COOH
c) Oleic acid — iii) C17H31COOH
A) a → i, b → ii, c → iii
B) a → iii, b ii, c → i
C) a → iii, b → i, c → ii
D) a → ii, b → iii, c → i ‘
Answer:
C) a → iii, b → i, c → ii
Question 122.
Alkaline hydrolysis of tri esters of higher fatty acids producing soaps is called
A) esterification
B) hydrolysis
C) saponification
D) neutralization
Answer:
C) saponification
Question 123.
Ravi added acid to the metal hydrogen carbonate and observed the gas evolved. The evolved gasis
A) O2
B) N2
C) H2
D) CO2
Answer:
D) CO2
Question 124.
The carboxylic acid used to preserve pickles is
A) Methanoic acid
B) Propanoic acid
C) Ethanoic acid
D) Butanoic acid
Answer:
C) Ethanoic acid
Question 125.
CH3 — CH2 — OH reacts with alkaline KMnO4 and heats to form first the compound A, which futher oxidises to form a compound B. The name of compound B is
A) Ethanol
B) Ethanal .
C) Formaldehyde
D) Acetic acid
Answer:
D) Acetic acid
Question 126.
But – 2 – yne reacts with H2 in the presence of ‘NT catalyst to form But -1 – ene. This reaction is an example of ………………….
A) substitution reaction
B) addition reaction
C) elimination reaction
D) rearrangement reaction
Answer:
B) addition reaction
Question 127.
Which of the following organic compounds undergoes substitution reaction ?
A) Alkanes
B) Alkenes
C) Alkynes
D) All
Answer:
A) Alkanes
Question 128.
Which of the following substitution products is not formed when methane reacts with chlorine in sunlight ?
A) chloroform
B) carbon chloride
C) methylene chloride
D) ethyl chloride
Answer:
D) ethyl chloride
Question 129.
The process of conversion of starch and sugar into ethanol by using enzymes is called
A) Fermentation
B) Esterification
Q Carbonization
D) Pyrolysis
Answer:
A) Fermentation
Question 130.
The sweet odour substance formed by the reaction of an alcohol and carboxylic acid is
A) ester
B) amine
C) ether
D) aldehyde
Answer:
A) ester
Question 131.
The general formula of alcohol is
A) CnH2n+1OH
B) CnH2n+1NH2
C) CnH2n+1CH0
D)(CnH2n+1)2O
Answer:
A) CnH2n+1OH
![]()
Question 132.
In combustion of alkane the heat energy is
A) absorbed
B) liberated
C) neither absorbed nor liberated
D) depends on alkane
Answer:
B) liberated
CONCEPT VI: Soap – Saponification Micelle
Question 133.
A spherical aggregate of soap molecule in water is called …………….
A) nucleus
B) cell nucleus
C) micelle
D) none of these
Answer:
C) micelle
Question 134.
CMC means
A) Critical Molar Concentration
B) Conical Molar Concentration
C) Critical Micelle Concentration
D) Conical Micelle Concentration
Answer:
C) Critical Micelle Concentration
Question 135.
In true solution the solute particle diameter is
A) more than 1 nm
B) less than 1 nm
C) equal to 1nm
D) none of these
Answer:
B) less than 1 nm
Question 136.
The polar end in micelle ……………… in nature.
A) hydrophilic
B) hydrophobic
C) isochoric
D) isothermal
Answer:
A) hydrophilic
Question 137.
The non polar end in micelle is ……………. in nature.
A) hydrophilic
B) hydrophobic
C) isochoric
D) Isothermal
Answer:
B) hydrophobic
Question 138.
The carboxyl end in micelle is …………….. in nature.
A) polar
B) non- polar
C) both
D) none of these
Answer:
A) polar
Question 139.
The hydrocarbon chain in micelle is ……………. in nature.
A) polar
B) non-polar
C) both
D) none of these
Answer:
B) non-polar